 |
|
|
|
|
Research Projects:: Formation and Stability of Simple Quinone MethidesThe parent p-quinone methide 1 and its relatives that contain the quinone methide functionality have long attracted the interest of discerning chemists. 1 can be thought of as a formally neutral benzylic carbocation at which there is limiting resonance stabilization by electron donation from a p-oxygen anion substituent to the cationic benzylic carbon. This strong interaction results in a high kinetic stability of, and large nucleophile selectivities towards, quinone methides, and is responsible in part for the interesting biological activity observed for more complex quinone methides. 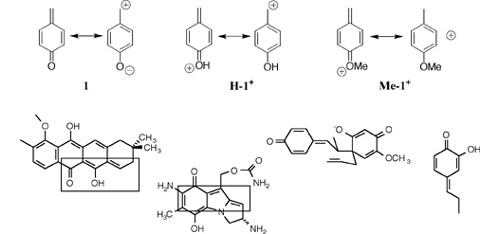
We have used two simple quinone methides as platforms to address several general questions about the effect of this very strong delocalization of charge on electrophilic reactivity. (1) We have shown by several criteria of their chemical reactivity that quinone methides behave as a member of the larger class of strongly resonance-stabilized carbocations, where resonance stabilization is provided by electron-donation from the 4-alkoxy substituent. See Journal of the American Chemical Society, 122, 1664-1774 (2000). 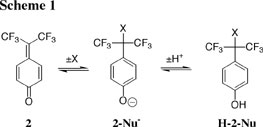
(2) There have been few measurements of rate and equilibrium constants for addition of halide ions to weakly reactive electrophiles, because these anions do not form stable adducts to such electrophiles. The adducts of halide ions to 2 (2-Nu, Scheme 1) are likewise unstable; however, rate and equilibrium data for their formation can be obtained by coupling nucleophile addition to protonation of the phenoxide oxygen of 2-Nu- to give H-2-Nu (Scheme 2). We have reported report rate and equilibrium data for the addition of halide and acetate ions to 1, which allows for extension to these nucleophiles of the well-known Ritchie N+ relationship for carbocation-nucleophile addition reactions. See Journal of the American Chemical Society, 122, 1664-1774 (2000). (3) We are interested in understanding how the transition state for nucleophile addition changes with extreme changes in electrophile reactivity. We have reported that the Broensted parameter βnuc = 0.11 for addition of strongly nucleophilic thiol anions to the weak electrophile 2, with rate constants kNu = 3 x 106 M-1 s-1, is significantly smaller than βnuc = 0.32 for addition of weakly nucleophilic alcohols to the 1-(4-methoxyphenyl)ethyl carbocation, with rate constants kNu = 1 x 107 M-1 s-1. This striking observation of similar rate constants, but very different values of βnuc, for two related carbocation-nucleophile addition reactions provides interesting insight into the nature of the reaction coordinate profiles for nucleophile addition. See Journal of the American Chemical Society, 122, 11073-11084 (2000). (4) We have used 2 in the first direct comparison of the reactivities of thiol anions (RS-), thiols (RSH) and sulfides (RSR) towards carbocations. Such a comparison is difficult because thiol anions are strongly nucleophilic and undergo activation-limited addition only to relatively stable carbocations, and these carbocations do not form stable adducts with the much more weakly nucleophilic sulfides. Our data show that the effect of substitution of Me for H at RSH on nucleophilic reactivity in water is a significant fraction of that expected for reaction in the gas phase, where the greater polarizability of the methyl group provides strong stabilization of positive charge at sulfur. See Journal of the American Chemical Society, 122, 11073-11084 (2000). 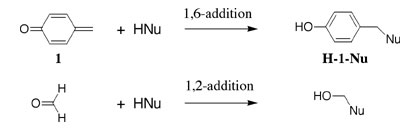
(5) We have compared rate and equilibrium date for 1,6 addition of water to the simple quinone methide 1 with the corresponding data for the 1,2-addition of water to the simple carbonyl group at formaldehyde. This comparison provides much insight into the effect of formation of the aromatic ring in the product of 1,6-addition on the thermodynamic driving force for addition of water to p-quinone methide 1. See Journal of the American Chemical Society, 125, 8814-8819 (2003). |
| |||||||||
All material © John Richard 2003-2004 | Website Developed by Iserloh Design | ||||||||||
