 |
|
|
|
|
Research Projects:: Formation and Stability of CarbocationsAs a postdoctoral fellow more twenty years ago I developed a general method now known as the "azide ion clock" for determination of the lifetimes of carbocations in aqueous solvents. Over the last twenty years, members of my group have applied this method to the determination of the lifetimes of a broad range of carbocations, including strongly-destabilized and strongly-stabilized carbocations; and so-called "antiaromatic" carbocations. The data have been analyzed to provide a clear description of many aspects of carbocation reactivity including: 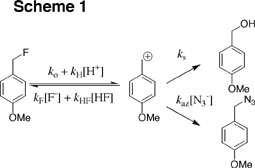
(1) Studies on the effects of aqueous solvation on carbocation reactivity has been examined. We have paid special attention to the strong solvation of fluoride ion which has the effect of entrapping this anion so that nucleophile addition to the 4-methoxybenzyl carbocation is barely detectable (Scheme 1). On the other hand, our work has shown that solvation of alkenes has little effect on the selectivity observed for these nucleophiles in aqueous solution. See (a) Journal of the American Chemical Society, 120, 10372-10378 (1998) and (b) Journal of the American Chemical Society, 124, 9798-9805 (2002). 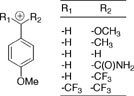
(2) A comprehensive characterization of the effect of changing carbocation structure on rate and equilibrium constants for carbocation formation. A wide range of substituents has been examined, ranging from strongly electron-donating (-OCH3) to strongly electron-withdrawing. The most surprising and revealing result of these studies is that the rate constants for nucleophilic addition to α-substituted methoxybenzyl carbocations are largely independent of the α-substituents at the benzylic carbon. See Journal of the American Chemical Society, 117, 5198-5205 (1995). (3) A detailed study of the formation and stability of tertiary carbocation intermediates of solvolysis at tertiary carbon in water. This work addressed the controversial question of the importance of nucleophilic solvation in providing stabilization of the transition state for solvolysis at tertiary carbon. A simple description for these reactions has been developed, which leaves minimal scope for controversy. Added insight into this reaction has been obtained through computational studies in the laboratory of Jiali Gao. See: Contemporary Reactive Intermediates, R. A. Moss, M. S. Platz, and M. Jones, Jr. ed. John Wiley and Sons: New York, pp 41 - 68 (2004). (4) A thorough examination of the effect of changing carbocation structure on the relative rate constants for carbocation partitioning between nucleophilic addition of solvent and loss of a proton from α-methyl carbon to form an alkene. See Advances in Physical Organic Chemistry, 35, 67-115 (2000). (5) A thorough characterization of the ambident nucleophilic reactivity of phenol and phenoxide ion (Scheme 2). See Journal of the American Chemical Society, 125, 15455-15466 (2003). 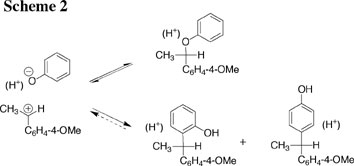
Many of the questions about carbocation reactivity addressed in this work have been answered, and many new questions have been raised so that this remains an area in which much exciting work is possible. One problem where considerable further progress is possibly concerns the characterization of the effect of sulfur bridging on the kinetic and thermodynamic stability of α-thioamide substituted carbocations. |
| |||||||||
All material © John Richard 2003-2004 | Website Developed by Iserloh Design | ||||||||||
