 |
|
|
|
|
Research Projects:: Mechanism of Enzymatic Catalysis of Proton Transfer: Triosephosphate Isomerase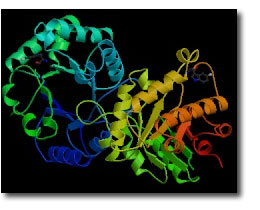
Triosephosphate isomerase (TIM) catalyzes a 1,2 proton shift at dihydroxyacetone phosphate (DHAP) to form D-glyceraldehyde 3-phosphate (GAP) through an enediol(ate) phosphate intermediate (Scheme 1). The enzyme's low molecular weight (46,000 daltons), high cellular abundance, and the centrality of proton transfer at carbon in metabolic processes have made TIM a prominent target for studies on the mechanism of enzyme action. Consequently, the emergence of new experimental methods for investigation of enzymatic reaction mechanisms, such as the use of tritium in tracer levels to monitor hydron transfer, Fourier transfer IR, NMR, X-ray crystallography, site-directed and random mutagenesis, computational modeling of enzyme catalysis and the design of transition state analog inhibitors can be traced through studies on TIM. This work has shown that TIM approaches perfection in its catalysis of triosephosphate isomerization, and it has provided a detailed description of the chemical events that occur at the enzyme active site. 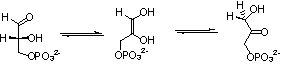
We are interested in understanding the mechanism by which this enzyme achieves a large rate acceleration for proton transfer. This rate acceleration is due to specific stabilization of the transition state for proton transfer by interaction with the protein catalyst. We have quantified this transition state binding energy by determining the free-energy reaction profiles for general base-catalyzed isomerization of GAP and for the TIM-catalyzed reaction, and then comparing these with the reaction profile determined by Knowles and coworkers for the TIM-catalyzed reaction of GAP. A comparison of these profiles shows that the transition state for the TIM-catalyzed reaction is stabilized by 16 kcal/mole relative to the transition state for the reaction catalyzed by the small buffer base quinuclidinone by interaction with the enzyme catalyst and that 14 kcal/mol (ca. 80%) of this transition state stabilization is provided by interactions between the enzyme and phosphate group of substrate. See (a) Journal of the American Chemical Society, 106, 4926-4936 (1984) and (b) Journal of the American Chemical Society, 123, 11325-11326 (2001). 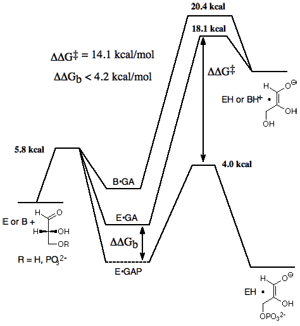
We are currently investigating the mechanism by which TIM utilizes the binding energy of the nonreacting portion of the substrate GAP to stabilize the transition state for proton transfer that occurs at a site distant from the phosphate group. Our working model is that this phosphate binding energy is used to move the substrate into a nonpolar environment in which the basicity of Glu-137 for abstraction of the substrate proton is enhanced, and electrostatic and hydrogen bonding interactions between the enzyme and the enediolate oxyanion are enhanced. The results of experiments to monitor TIM-catalyzed exchange between solvent and substrate, monitored by methods developed in our nonenzymatic studies of carbon acids, are consistent with the notion that isomerization occurs in a protected, nonpolar enzyme active site [in preparation]. Future work will involve the use of site-directed mutagenesis to probe the effect of specific interactions between the enzyme and phosphate group of substrate on catalytic activity. |
| |||||||||
All material © John Richard 2003-2004 | Website Developed by Iserloh Design | ||||||||||
