|
|
|
|
|
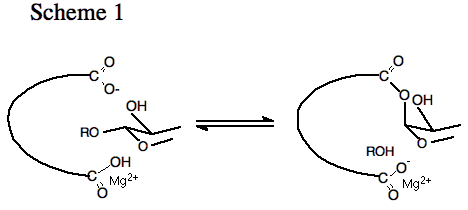
Research Projects:: Mechanism of Enzymatic Catalysis of Glycosyl Transfer: β-Galactosidase.Many of the details of the mechanism for enzymatic catalysis of glycosyl transfer reactions are not well understood. Scheme 1 shows a working model for the first step for catalysis of glycosyl transfer with retention of configuration, which was first proposed after examination of an X-ray crystal structure of a lysozyme-inhibitor complex. This proposal is now supported by extensive results of model studies of nonenzymatic catalysis of acetal cleavage.
This reaction mechanism is defined by the imperatives for catalysis of nucleophilic substitution of a poor alkoxide ion leaving group at glycosides, and these imperatives will probably ensure the presence of the following essential catalytic residues or metal cofactors at all enzymes that catalyze glycosyl transfer with retention of configuration at the glycosidic carbon: (1) A nucleophilic residue that participates either by providing assistance to expulsion of the leaving group from the anomeric carbon, and/or electrostatic stabilization of an oxocarbenium ion reaction intermediate. (2) An acidic residue (e.g., a carboxylic acid or a metal ion) that provides stabilization of negative charge at the oxygen leaving group. In the second step for the hydrolysis reaction, the residue that functions in catalysis of leaving group expulsion acts as a catalyst of the addition of water (ROH = HOH) to the glycosyl-enzyme intermediate. Our work on this enzyme has focused on several problems. 
(1) The characterization of the effect of changing alkyl alcohol leaving group/nucleophile on catalytic activity. See (a) Biochemistry, 34, 11703-11712 (1995) and (b) Biochemistry, 34, 11713-11724 (1995). (2) Site-directed mutagenesis studies that have provided insight into the mechanism for the enhancement of acid-base catalysis by β-galactosidase. See (a) Biochemistry, 37, 4305-4309 (1998) and (b) Bioorganic Chemistry, 8, 146-155 (2001). (3) The 28000-fold difference in the values of kS = 0.0046/s for hydrolysis of the 2-deoxy galactosyl-enzyme intermediate of β-galactosidase and 1300/s for hydrolysis of the intermediate of the physiological reaction, is consistent with at least a 7.4 kcal/mole stabilization of the transition state for the physiological reaction by interaction of β-galactosidase with the C-2 hydroxyl group. This binding interaction will be even greater than 7.4 kcal/mole, if the inductive effect of the C-2 hydroxyl group causes the same ca. 1000-fold reduction in the rate constant for sugar hydrolysis as observed for the physiological reaction. By comparison, it has been estimated that glycosyl transferases provide a ca. 20 kcal/mol stabilization of the transition state for cleavage of simple glycosides, which corresponds to a 20 kcal/mol transition state binding energy. In the case of b-galactosidase these binding interactions involve largely the protein and the β-D-galactopyranosyl group of substrate because this enzyme is an efficient catalysis of cleavage of alkyl β-D-galactopyranosides, where the leaving group is a small alkyl alcohol such as ethanol and trifluoroethanol. The large fraction of this transition state stabilization that originates in some manner from interaction with a single C-2 hydroxyl group is striking, but poorly understood and is currently under investigation in our laboratory. |
| |||||||||
All material © John Richard 2003-2004 | Website Developed by Iserloh Design | ||||||||||