 Wayne K. Anderson
Wayne K. Anderson  Wayne K. Anderson
Wayne K. Anderson
Dean, School of Pharmacy and Pharmaceutical Sciences
Professor of Chemistry
B.Sc. Pharmacy (University of Manitoba); M.Sc. Pharmaceutical Chemistry (University of
Manitoba);
Ph.D. Medicinal Chemistry (University of Wisconsin at Madison)
Research Interests and Selected Recent P ublications
Drug design; prodrugs; natural product synthesis; new synthetic methods; heterocyclic chemistry.
The primary focus of the research in our laboratory is the design and synthesis of novel chemotherapeutic agents with potential utility in the treatment of cancer or AIDS. Three general approaches are used: (1) drug design based on novel "lead" structures; (2) design of enzyme inhibitors, and; (3) design of DNA targeted agents.
"Lead" structures are selected on the basis of distinctiveness, possible novel mechanism(s) of action, and/or potential for significant innovation. We use naturally occurring alkaloids, terpenes, marine toxins, and antibiotics as "lead" structures for drug design. We also use platinum diamine complexes, DNA cross-linking agents, and DNA minor groove binding agents as "lead" compounds. One challenge, once a "lead" structure is identified, is the development of specific structural hypotheses that can be tested in drug designs. We use computational chemistry, molecular modeling, and quantitative structure activity relationship correlations ("QSAR") as tools to develop and refine drug design hypotheses; X-ray crystallographic data and NMR data are often used in conjunction with these studies. The synthesis of the newly designed compounds is also a major challenge, and, often synthetic routes are designed to permit maximal structural divergence.
Several different projects are currently in progress.
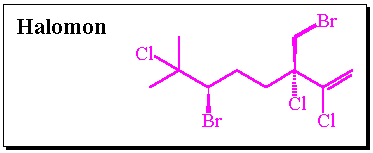
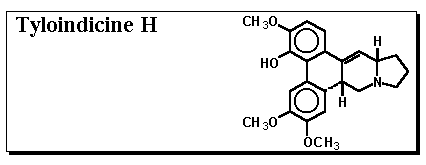
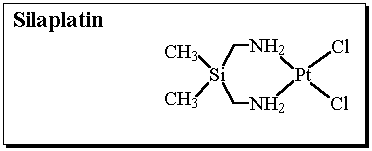
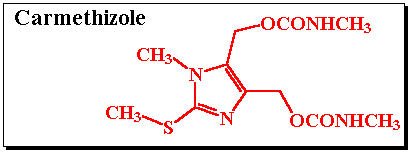
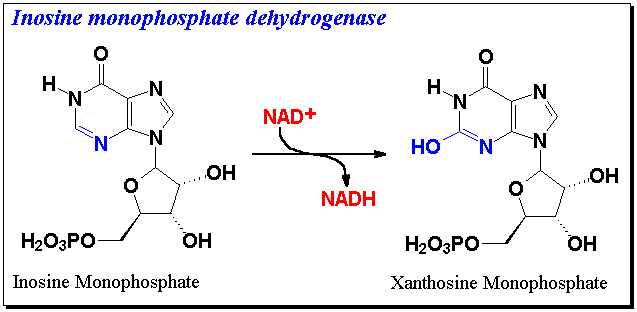
Inosine monophosphate dehydrogenase (IMPD) catalyzes the conversion of inosine monophosphate (IMP) to xanthosine monophosphate (XMP) and is the rate limiting enzyme involved in the de novo biosynthesis of guanine nucleotides.
Wayne K. Anderson wka@buffalo.edu